iStock.com/inkoly
Understanding the importance of normal pH levels in the body first requires understanding what the pH scale represents and how it affects the body’s functions.
Body systems require a level of both acidity and alkalinity to protect against disease and promote healing.
Too much, or too little, of either can be damaging to various physical and mental activities.
In This Article:
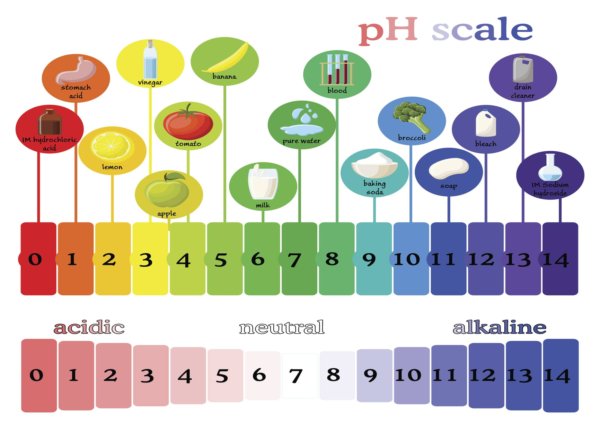
What is pH balance?
The term “pH” refers to the potential of hydrogen within substances. A normal, or healthy, measurement for the human body is about 7.4 on a scale ranging from 0 to 14. The closer to 0 the body’s pH reading, the more acidic it is, while a reading leaning towards 14 reflects a more alkaline state. A pH of 7 is neutral; therefore, the optimal reading is slightly alkaline.
pH Levels in the Body and Its Role
At no one time are pH levels uniform throughout the body, however, as many biochemical processes necessitate consistent acid production. Acid levels can also change as a result of food choices. The modern Western diet, for example, is filled with foods that tend to promote high levels of acidity within the body.
The pH levels throughout the body vary, with the highest acid content existing in the digestive system, mainly the stomach. The stomach maintains a pH of 1.35 to 3.5 due to gastric acid, which helps the organ break down proteins and fight off invading pathogens.
While the lining of the stomach is more alkaline to protect against injury, any decrease in the barrier’s acid-neutralizing secretions may lead to duodenal ulcers in some.
Shifts in pH will have differing effects on health, depending on the location within the body.
The more acidic pH level of the skin protects against the overgrowth of microbial invaders, and ranges between 4 and 6.5.
The pH of urine is beneficial at a level of 4.6 to 8.0.
Like gastric acid, digestion aids such as bile and pancreatic fluid break down nutrients, but they also neutralize stomach acid. Bile has a pH of 7.6 to 8.8, while that of pancreatic fluid is 8.8 on average.
Vaginal fluid retains a pH of less than 4.7 to limit an abundance of harmful microorganisms, and the brain’s exterior is protected by a cerebrospinal fluid pH of 7.3.
An average pH of 6.0 to 7.2 of intracellular fluid is necessary due to the production of acid within the cells.
Overall, the human body must maintain a pH range of 7.35 to 7.45 for survival. What is considered “normal” or healthy for blood is 7.4, and the body works diligently to uphold the balance. Breathing is one method, as the body controls acid levels by expelling carbon dioxide with each exhale. Acid can also be discharged through the urine, skin, and other body fluids.
How an Unbalanced pH Affects the Body
Having excess or deficient levels acidity can affect every system within the body, as well as its physical structures. Restoring the pH balance in the body sometimes requires the body to draw from a stored base supply of carbonates and phosphates (usually in the bones), which can negatively affect proper functioning of these systems and structures.
Acidosis, the presence of excess acid in the body’s fluids, is a major factor in stroke, diabetes, heart disease, osteoporosis, arthritis, fibromyalgia, multiple sclerosis, and cancer. An underlying common thread is the inflammation that may accompany unbalanced pH levels.
Some experts believe that the common symptoms of muscle pain, breathing difficulty, and constant fatigue are the first signs of excess acidity. Symptoms of unbalanced pH levels will depend on the source.
Circulatory system: Inflammation is an underlying factor in heart disease. A surplus of acid can add to cardiovascular issues as the tissues become inflamed and fats are then used to protect the lining of the arteries. These conditions can lead to heart failure as the blood is unable to flow easily to and from the heart.
Digestive system: For proper functioning of the digestive tract, it’s important to replace the excess acid in the standard Western diet with an alkaline-based diet (rich in fruit and vegetables, seeds, and nuts). Insufficient alkaline levels can cause nausea, bloating, and gastric reflux as the pancreas fails to send signals to the other organs, resulting in inflammation.
Urinary system: Encompassing the kidneys, the ureter, the urethra, and the bladder, the urinary system can facilitate the loss of alkaline minerals in the presence of high-acid levels. The system will draw alkaline from the bones, tissues, and organs and transfer it into the bloodstream to compensate, which can lead to excruciating kidney stones.
Immune system: Maintaining a balanced pH level could serve to direct pathogens away from the immune system. High levels of acid may lead the microorganisms to settle in hidden areas. Modern medical treatments such as drugs, surgery, and chemical radiation can hinder the body’s natural healing response.
Integumentary system: This system comprises the skin, nails, hair, and exocrine glands. Acidosis affects the conditioning of the skin’s tissues by prompting a buildup of acid deposits. The resulting inflammation impairs the skin’s ability to block infections and invading pathogens. This can lead to pimples, rash, wounds, sores, and lesions.
Muscular system: Energy is required for the proper development and functioning of the muscular system. The body breaks down oxygen and glucose metabolically to generate this energy. Acidosis can disrupt the metabolic processes that nourish cells, leading the muscles to perform poorly.
Nervous system: The nervous system also depends on constant energy for physical, mental, and emotional strength. The high levels of acidity may hinder this process.
Reproductive system: While much more research is needed to fully understand the connection between infertility and sexual dysfunction and acidosis, preliminary studies suggest that excess acidity is linked to decreased arousal, sexual enjoyment, and fertility.
Respiratory system: As noted, oxygen supply to the cells may be hindered by excess acidity. A buildup of toxic waste and mucus in the lungs and other areas of the respiratory system could potentially lead to asthma, bronchitis, and recurrent colds.
Skeletal system: Acidosis may cause inflammation to settle into the joints, leading to arthritis symptoms of stiffness and pain. A buildup of large uric acid deposits within the joint cells results in painful crystals in commonly used joints such as the wrists, knees, and feet.
For patients with chronic kidney disease, or CKD, high-acid diets might increase the risk of kidney failure. Results of a study indicate that patients with CKD can greatly benefit by improving their diet to include more fruits and vegetables, and by eating less meat and processed foods. The research team found that high levels of acid in the diets of patients with CKD was closely linked to developing kidney failure at a rate three times faster than those consuming a low-acid diet.
When you eat a highly acidic diet (one in which meat and processed foods heavily outweigh fruits and vegetables), it becomes harder for your body to repair damaged cells and absorb nutrients, and you become more susceptible to disease and illness. These problems are exacerbated in people with chronic kidney disease because they lead to a buildup of waste in your blood.
Restoring pH Balance in the Body
A pH balance is the ratio of acidity to basicity. In humans, one of the most important factors in determining pH levels is diet. Eating too many acidic foods can lead to conditions like diabetes and chronic kidney disease, among others.
Ideally, you want to include 60% alkaline-forming foods in your diet and 40% acid-forming foods.
Again, because of the standard American diet, it is very easy to have an acidic pH. Processed foods containing added sugar and salt are typically always high-acidity options, as are most meats and poultry.
On the other hand, most fruits and vegetables are alkaline. Striking a healthy balance between the two is essential for your health, relieving stress from your organs and the various systems working inside you that are essential to life.
Simply put, be aware of the pH factors of foods and drinks you consume. The healthier and more natural the food, the more alkaline it likely provides. In contrast, the less healthy a food is considered, the greater the chance it exceeds the preferred acidity level. For example, soda is a 2 on the pH scale, while green veggies are 7 or above.
Importance of Restoring pH Levels
The importance of restoring the pH level to an acceptable level can be seen in the effects of the acidity and alkaline on the body and certain medical conditions.
Sarcopenia
An unbalanced pH level can exacerbate the loss of muscle mass that naturally occurs over time. Weak muscles can lead to bone fractures from falls and reduced mobility. The effects of the extra acid are particularly evident in conditions such as sepsis, renal failure, ketosis, chronic obstructive lung disease, diabetic ketosis, and trauma to the muscle.
Growth Hormones
With age, the natural production of growth hormones slows and negatively affects cell regeneration. High levels of acidity can rapidly impair hormone production. The growth hormones are vital to the musculoskeletal system, the cardiovascular system, and cognitive behavior.
Pain
The inflammation that leads to pain may be triggered by the consumption of foods high in acidity. Processed, high-fat, and junk foods appear to be most damaging. The alkaline content in healthy foods could potentially adjust the pH balance to a more stable state by detoxifying the body.
Chemotherapy
Cancer treatments such as chemotherapy require the pH level to be balanced for full effectiveness. Depending on the agents used, a more acidic or a more alkaline-based agent is a main component of the treatment.
Restoring pH Balance with Diet and Lifestyle Changes
You can make certain changes to help your body’s pH levels return to normal.
- Add chlorophyll-based foods and drinks to your daily diet. Dark, green leafy vegetables like spinach and kale, herbs like dill and parsley, and sea vegetables offer both immune-boosting and detoxifying properties.
- Use lemons to alkalize the body. In natural form, lemon can be acidic, but when combined with water, it becomes a source of mineral salt compounds. Squeeze one-quarter of a fresh lemon into two cups of lukewarm water for refreshing drinks throughout the day.
- Consume root vegetables such as onions, leeks, sweet potatoes, carrots, and beets to better absorb calcium through their inulin content. Inulin supports good gut bacteria to help lower pH levels.
- Stay away from refined sugar-based products as the body’s stored alkaline minerals are needed to safely excrete the highly acidic urine these foods and beverages can produce. This includes soda, aspartame-rich products, and fructose additives.
- Choose protein from plants rather than animals. Limit red meat intakes to four ounces per serving.
- Exercise regularly and practice proper breathing techniques to help relieve tension, anxiety, and stress, which trigger the release of hormones that produce high levels of acidity.
- Avoid smoking as tobacco contains chemicals that significantly raise acid levels within the body.
For a clearer picture of a pH diet, refer to the sample meal plan below:
- Breakfast: Two scrambled eggs mixed with onions, tomatoes, and spinach; ginger tea
- Snack: Pear, toasted pumpkin seeds
- Lunch: Grilled salmon; salad of mixed greens, tomatoes, carrots, cucumber, and broccoli
- Snack: Almonds
- Dinner: Baked chicken, sweet potato, green salad
pH Balance Food Chart
The below pH balance chart can help you determine the pH levels of popular food choices.
| pH Level | Alkaline/Acidity balance | Foods |
| 1 | Acidic | Soda, energy drinks |
| 2 | Acidic | Cranberry juice, artificial sweeteners |
| 3 | Acidic | Processed food, chocolate, pork |
| 4 | Acidic | Coffee, beef, white bread |
| 5 | Acidic | Poultry, sugar, beer |
| 6 | Less Acidic | Grains, milk, cold-water fish |
| 7 | Neutral | Tap water, river water |
| 8 | Alkaline | Apples, mushrooms, wild rice |
| 9 | Alkaline | Sweet potato, grapes, olive oil |
| 10 | Alkaline | Spinach, carrots, cucumbers |
Test Your Body’s pH Levels
Keeping track of your body’s pH levels is as important as knowing the acidity of the foods you consume. A pH test is available at most pharmacies and large box department stores. While any bodily fluid can be used, testing the saliva of the mouth will give the most accurate reading.
A precise measurement can be obtained first thing in the morning or two hours after eating. Certain health conditions can affect your pH balance, such as cancer and mineral deficiencies.
First, swallow saliva. Once it gathers in the mouth again, spit it out. Then, spit newly gathered saliva onto the colored portion of the litmus paper. The color of the paper will change to reflect the acidity level. Use the color key code included with the kit to determine the pH level results.
It should be noted that if the color of the paper turns blue, the pH level is 7.4, a normal state.
Also read:
Article Sources (+)
Schwalfenberg, G.K., “The Alkaline Diet: Is There Evidence That an Alkaline pH Diet Benefits Health?” Journal of Environmental and Public Health, 2012; 2012:727630; https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3195546/, last accessed April 5, 2018.
“How Our Bodies Go To Extraordinary Lengths To Maintain Safe pH Levels,” Forbes, March 11, 2016; https://www.forbes.com/sites/quora/2016/03/11/how-our-bodies-go-to-extraordinary-lengths-to-maintain-safe-ph-levels/#29a02a6e70e5, last accessed April 5, 2018.
“pH balance in the body,” Women’s Health Network; https://www.womenshealthnetwork.com/digestivehealth/ph-balance-in-the-body.aspx, last accessed April 5, 2018.
“pH 101: Acid-Alkaline Balance & Your Health,” Kris Carr; http://kriscarr.com/blog/ph-balance-alkaline-foods/, last accessed April 6, 2018.
“How to eat a PH-balanced diet,” Body and Soul, June 17. 2016; http://www.bodyandsoul.com.au/diet/diet-az/how-to-eat-a-phbalanced-diet/news-story/e9fcc2b1c7fd42514e76af9064e7e959, last accessed April 6, 2018.
